Glutathione, Kojic Acid and Arbutin: What Is Truly Stable and What Is Legally Permitted?
Brightening ingredients are some of the most misunderstood components in cosmetic formulation. While many actives are heavily promoted for their ability to lighten, even skin tone, or reduce the appearance of pigmentation, far fewer resources explain what actually remains stable in real formulations, and what is legally permitted across different regulatory regions.
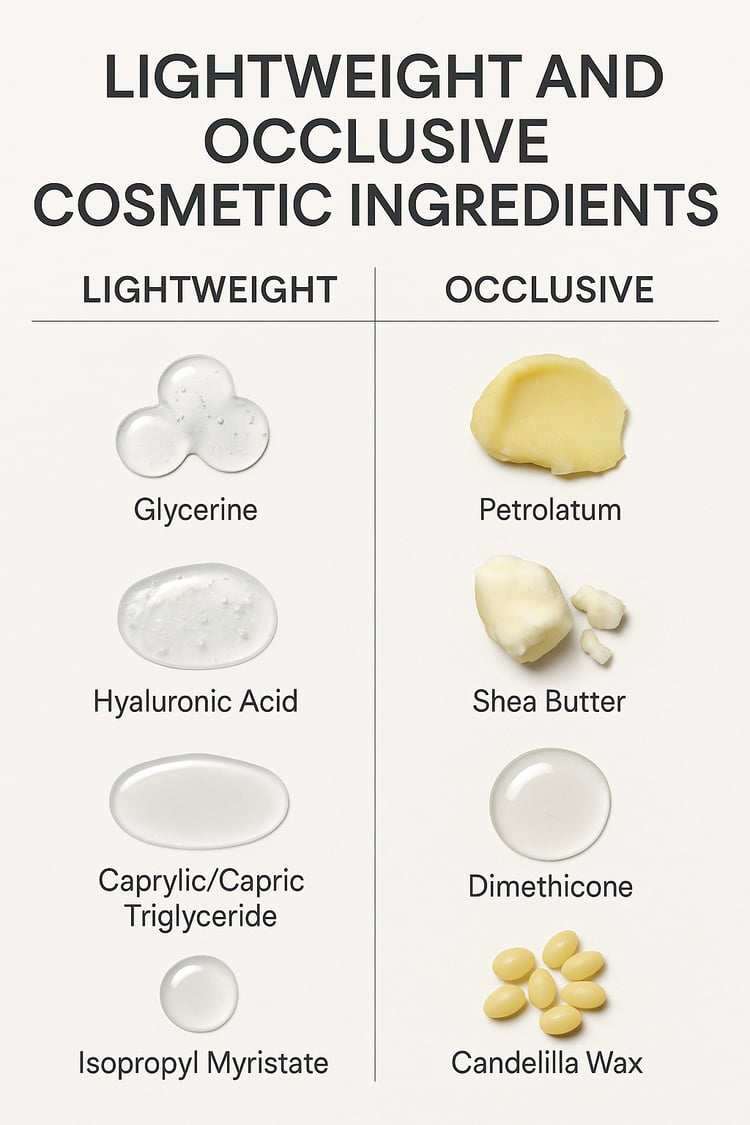
This educational document explores three of the most commonly requested brightening ingredients in modern skincare: glutathione, kojic acid, and arbutin, including alpha arbutin. Rather than focusing on marketing claims, this guide breaks down how each ingredient behaves in cosmetic systems, why stability issues commonly occur, and how formulation choices directly impact performance and shelf life.
You will also gain clear, up to date regulatory insight for Australia, New Zealand, the United States, and the European Union, including the latest EU concentration restrictions and compliance expectations. The document explains how product claims influence legal classification, why certain brightening products cross into therapeutic or drug categories, and how to design formulations that remain compliant across multiple markets.
This resource is written for formulators, brand owners, and skincare educators who want scientifically accurate, practical guidance rather than oversimplified ingredient trends. Whether you are developing brightening serums, creams, or treatment products, this guide will help you understand what truly works, what remains stable, and how to avoid costly regulatory mistakes.
If you are looking to create effective brightening formulations that hold up in real world conditions while staying legally compliant, this document provides the clarity every formulator needs.