ERP Template - ADO Config (CSV Example)
Azure DevOps Configuration Guide for CSV DOCX
ERP_Sherpas_ADO_Config_Guide.docx
A detailed, step-by-step configuration guide for setting up Microsoft Azure DevOps as a GAMP 5-aligned Computer System Validation platform for a Business Central ERP implementation in a pharmaceutical or regulated life sciences organisation. The guide covers GAMP 5 category classification for BC, regulatory context (EU Annex 11, ICH Q10), Azure DevOps project structure, process template customisation, custom GxP field definitions, area paths, permission matrix, test plan configuration for IQ/OQ/PQ phases, RTM query design, and evidence export.
For a PM delivering an ERP project in a regulated industry, this guide eliminates the need to design a bespoke validation platform from scratch — reducing both the risk of regulatory non-compliance, and will also support the internal CSV compliance team in designing and implementing the ADO setup for the project.
Azure DevOps for GxP Validation — Overview Presentation PPTX
ERP_Sherpas_ADO_GxP_Validation_Overview.pptx
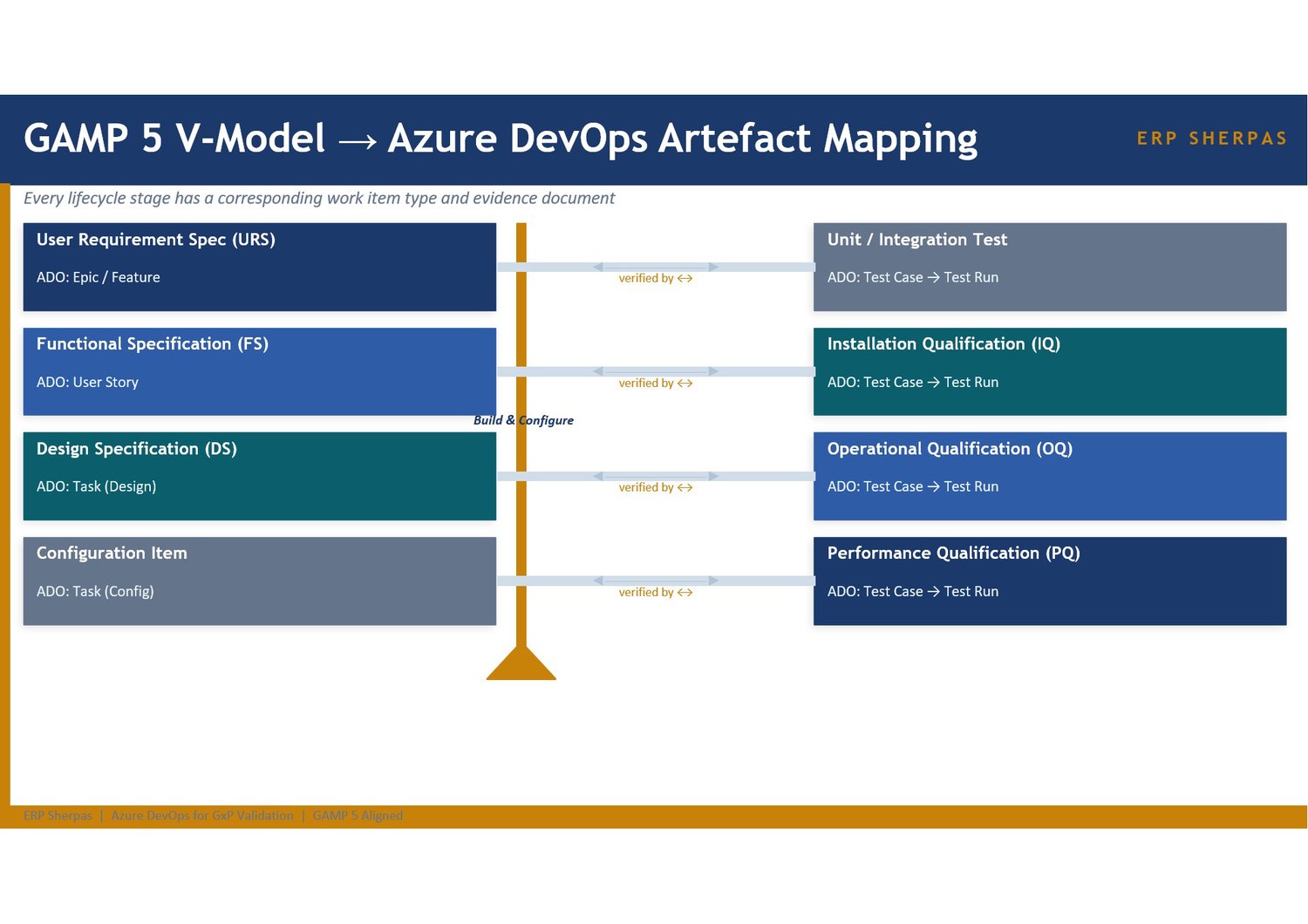
A 14-slide executive and technical overview presentation explaining how Azure DevOps, when correctly configured, provides a single auditable platform for all GAMP 5 V-model activities during a pharmaceutical ERP implementation. It covers the regulatory case for a centralised platform, the mapping of GAMP 5 V-model stages to Azure DevOps artefact types, recommended project structure, work item hierarchy and custom GxP field design, three fully worked traceability examples (Inventory lot tracking, Production BOM control, and Quality Management non-conformance), the CSV evidence pack ADO produces, audit trail and access controls, and a phased setup roadmap.
For a PM, this presentation makes the regulatory technology case to a client QA or IT audience — building confidence that ADO is a validation-grade platform.
GAMP 5 Requirements Traceability Matrix — Pharma XLSX
ERP_Sherpas_ADO_RTM.xlsx
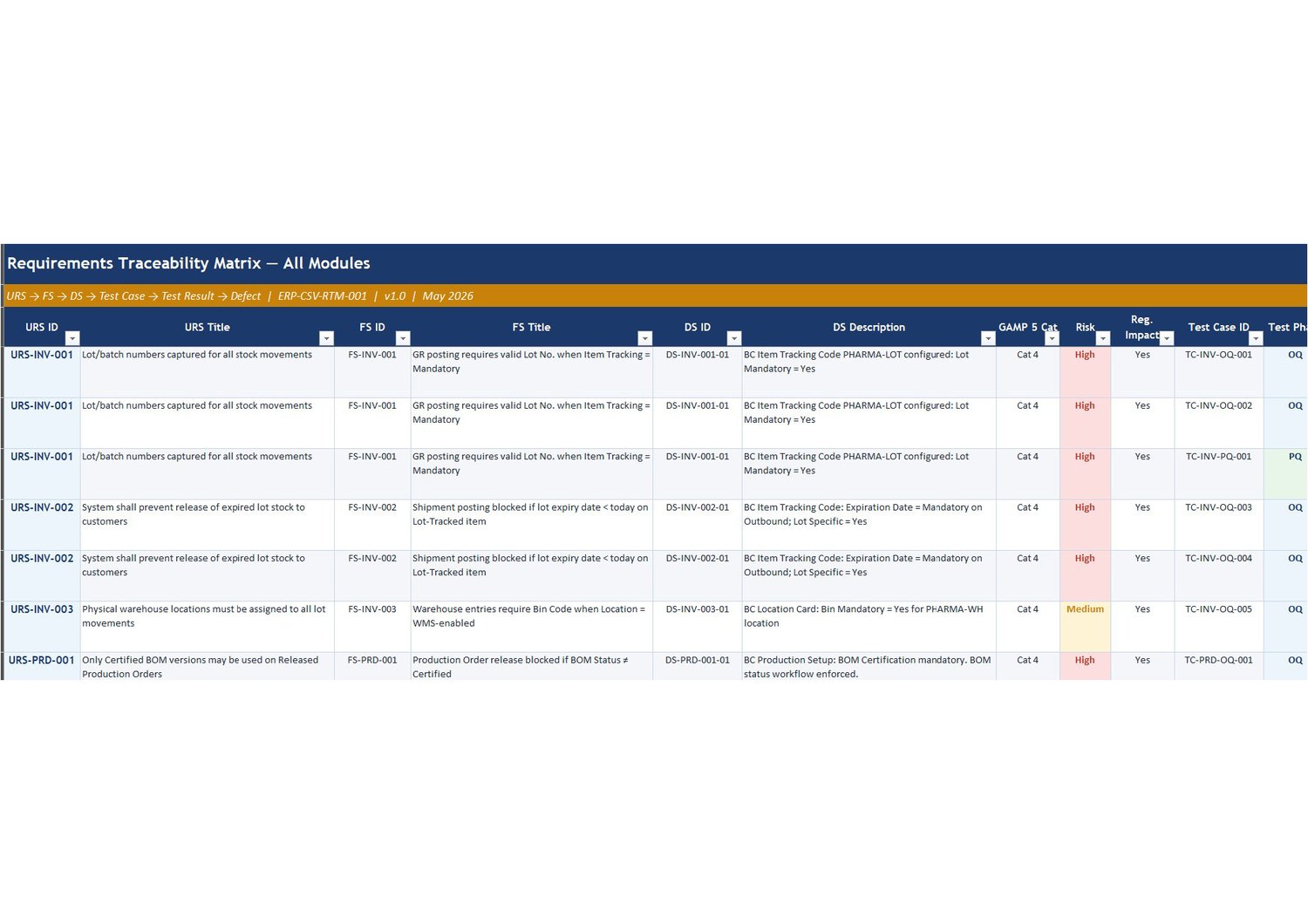
A fully populated worked example of a GAMP 5-aligned Requirements Traceability Matrix for a Business Central pharmaceutical ERP implementation, with seven sheets covering the complete URS→FS→DS→Test Case→Test Result→Defect traceability chain across three modules (Inventory and WMS, Production and MRP, and Quality Management), plus a test execution summary with IQ/OQ/PQ pass rates, a change control log, and an ADO work item reference guide. The matrix contains 19 fully traced requirements with real-world examples including lot traceability, BOM certification control, approved vendor list enforcement, and non-conformance CAPA linkage.
For a PM, this document is the difference between a validation project that passes a regulatory inspection and one that fails on traceability gaps — and having a real, populated example to baseline from eliminates months of framework design.