EU MDR Clinical Evaluation & PMS Pack
On Sale
€250.00
€250.00
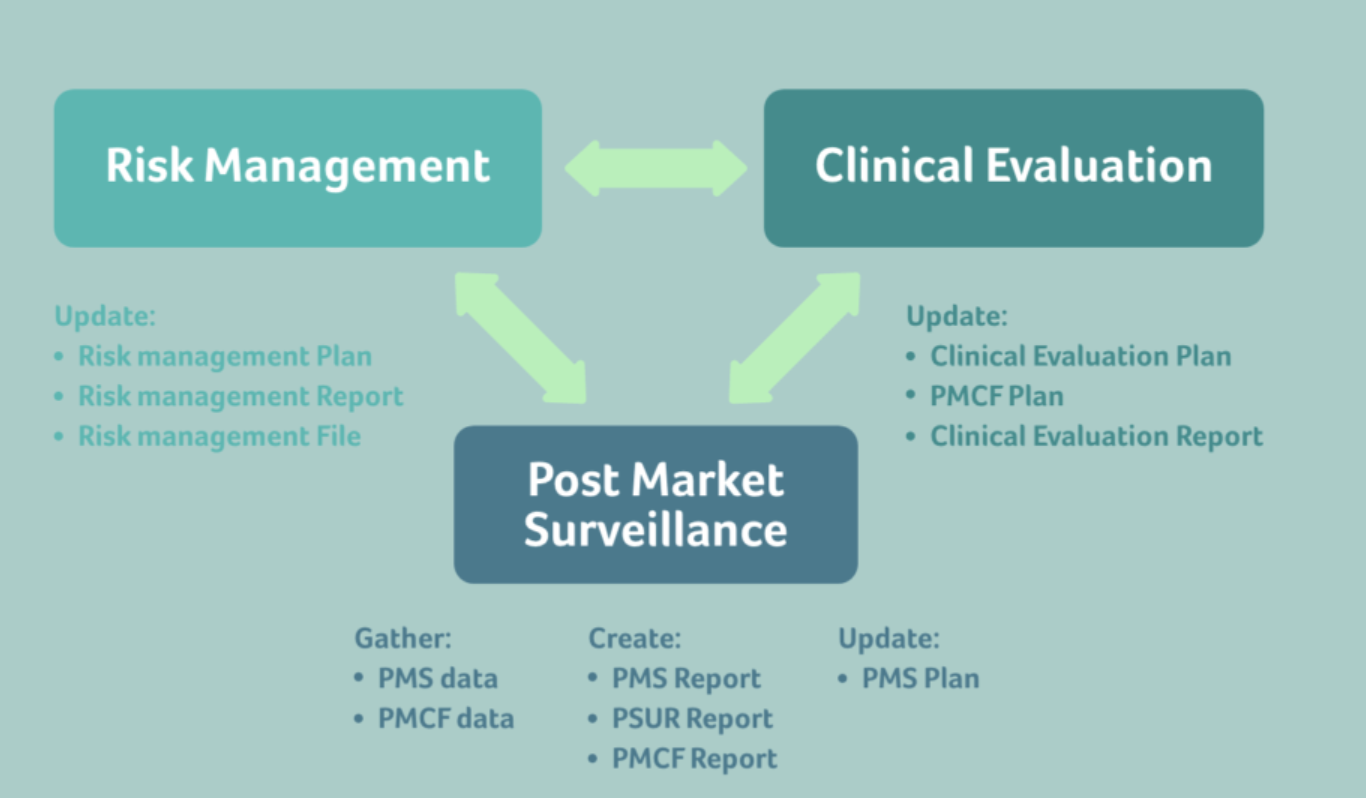
Clinical evaluation is a critical part of your medical device development process. Do not leave it to after you have placed your product on market before considering these requirements. Planning is essential for clinical data and post-market surveillance. This pack of document swill help you get started. Let us know if you need additional support.
This pack contains:
- Clinical Evaluation and Post-Market Clinical Follow-up SOP
- Post Market Surveillance SOP
- Vigilance Incident Reporting SOP
- Clinical Evaluation Report Template
- Post-market Surveillance Plan Template
- PMCF Plan and Report Template

