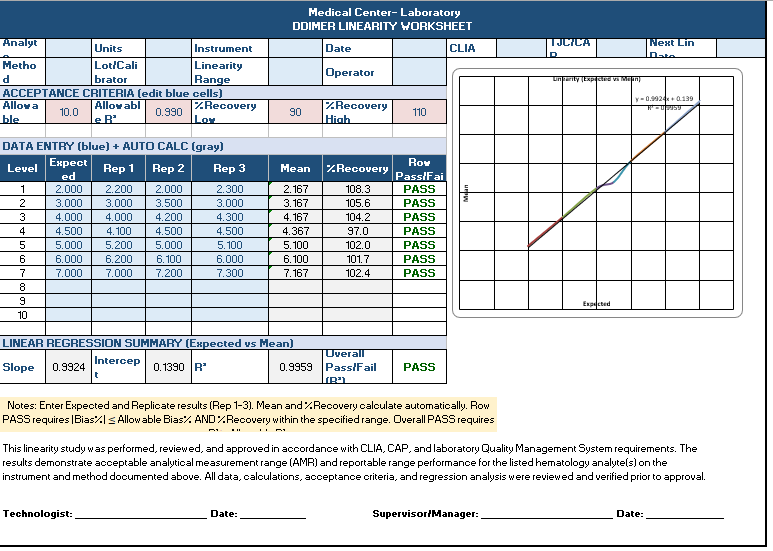
Linearity Worksheet
Medical Laboratory Linearity & Reportable Range Validation Worksheet
This Linearity & Analytical Measurement Range (AMR) Validation Worksheet is a professional, audit-ready laboratory tool designed to document and verify assay linearity, accuracy, and reportable range for any quantitative laboratory test.
Built to meet CLIA, CAP, and Joint Commission (TJC) quality system expectations, this worksheet provides a complete, single-page framework for evaluating precision, recovery, bias, and linear regression using replicate patient or calibrator dilutions.
Key Features
1. Fully Automated Calculations
The worksheet performs all required calculations automatically:
- Mean from three replicates
- % Recovery vs expected value
- Bias and deviation
- Pass/Fail per level
- Linear regression (slope, intercept, R²)
- Overall validation status
All calculation fields are protected to prevent accidental modification.
2. CLIA-Compliant Acceptance Criteria
Built-in regulatory thresholds include:
- Bias limits
- % Recovery limits
- Minimum R² requirement
- High and low recovery cutoffs
Each dilution level is automatically evaluated and flagged PASS or FAIL using regulator-defensible rules.
3. Single-Page Visual Linearity Graph
The worksheet generates a real-time Expected vs Mean plot with:
- True linear regression line
- R² and equation displayed on the chart
- Visual confirmation of assay linearity
Only one clean graph is shown, matching inspection and validation best practices.
4. Inspection-Ready Format
The layout mirrors how validation evidence is expected to appear during inspections:
- Analyzer and method identifiers
- Reagent and calibrator tracking
- Operator and date
- Acceptance criteria table
- Results table with pass/fail logic
- Final approval section
This format supports:
- CLIA inspections
- CAP checklist documentation
- Joint Commission tracer activities
- Internal audits
5. Professional Validation Report
The worksheet includes:
- Formal summary statement
- Technologist and supervisor sign-off
- Date fields
- Audit-ready layout
It can be printed, exported to PDF, or stored directly in your Quality Management System (QMS) or validation files.
Who This Is For
Designed for:
- Hospital laboratories
- Reference laboratories
- CLIA-waived to high-complexity testing
- Core labs and point-of-care programs
- New analyzer validations
- Lot-to-lot verification
- Ongoing CAP and TJC compliance
What You Get
A ready-to-use, protected Excel worksheet that allows you to:
- Enter expected values and replicate results
- Instantly evaluate assay linearity and AMR
- Generate regulator-ready validation documentation
No macros. No add-ins. Just clean, compliant, defensible data.

